Refractometers are measuring instruments for the refractive index of liquids and thus also for the sugar or alcohol content:

 On the first picture you can see a cheap hand refractometer.
There are some for sugar solutions and some for alcohol solutions.
They differ in the labeling of the scale and possibly also in the measuring range of the refractive index.
They are often equipped with "ATC", an automatic temperature compensation.
On the first picture you can see a cheap hand refractometer.
There are some for sugar solutions and some for alcohol solutions.
They differ in the labeling of the scale and possibly also in the measuring range of the refractive index.
They are often equipped with "ATC", an automatic temperature compensation.
The second picture shows an Abbe refractometer, a much more expensive professional laboratory instrument. This usually has a scale for the refractive index and one for the sugar content in Brix. However, the alcohol content can also be measured with it. You need a table or calculator for this to infer alcohol strength from refractive index. A temperature compensation is not built in here. This would also be nonsensical, since it is supposed to be a universal measuring instrument, and you would need a different compensation for each substance to be measured. Instead of correcting the measured value depending on the temperature, the measuring head is normally brought to the desired temperature by a temperature-controlled water circuit. Then one is also independent of the availability and accuracy of temperature correction tables or calculators for the respective substance.
The refractive indices of sugar and alcohol solutions
The relationship between refractive index and sugar concentration is quite linear: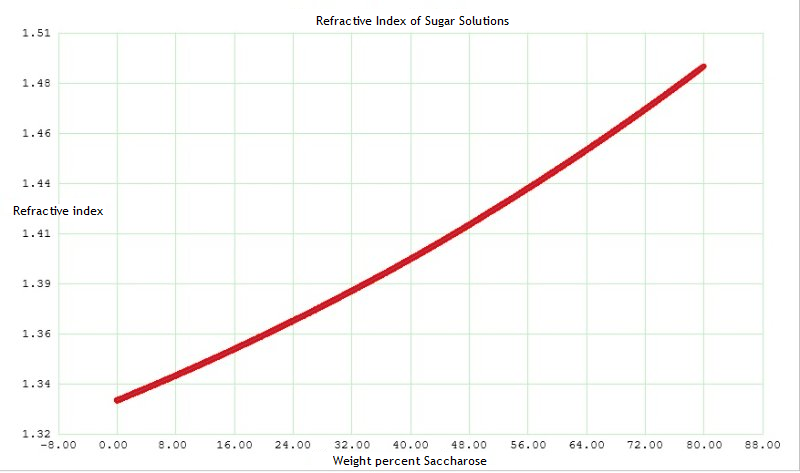 In contrast, with alcohol solutions:
In contrast, with alcohol solutions:
 The curve of alcohol solutions is not only much more complex, it even reverses in the upper alcohol strength range.
This means that there are two possible alcohol strengths for some measured refractive indices.
Thus, a measured index of 1.365 can mean either 75.3 or 93.2%abv.
The highest refractive index is at 1.3658 or according to our data at 85.7%abv.
The range where there are two alcohol strengths per refractive index is from 1.3614 - 1.3658 or 100 - 57.2%abv.
This is illustrated by our calculator Refractometer at high Alcohol Strength.
This calculator should make it possible to measure alcohol strengths close to the azeotrope.
The scale of alcohol refractometers goes because of the inversion of the curve only up to 80%abv:
The curve of alcohol solutions is not only much more complex, it even reverses in the upper alcohol strength range.
This means that there are two possible alcohol strengths for some measured refractive indices.
Thus, a measured index of 1.365 can mean either 75.3 or 93.2%abv.
The highest refractive index is at 1.3658 or according to our data at 85.7%abv.
The range where there are two alcohol strengths per refractive index is from 1.3614 - 1.3658 or 100 - 57.2%abv.
This is illustrated by our calculator Refractometer at high Alcohol Strength.
This calculator should make it possible to measure alcohol strengths close to the azeotrope.
The scale of alcohol refractometers goes because of the inversion of the curve only up to 80%abv:
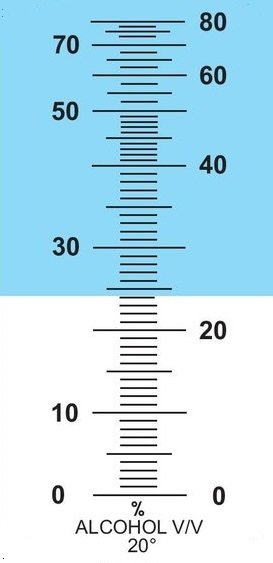 This scale also shows very clearly that in the range above 50%abv it becomes increasingly difficult to read off an accurate result.
In the range of 80%abv it is then so inaccurate that the use of a refractometer no longer makes sense.
And on the other side of the reversal point it is unfortunately not ideal either.
A refractometer is therefore more suitable for alcohol solutions when a low alcohol strength is expected.
This scale also shows very clearly that in the range above 50%abv it becomes increasingly difficult to read off an accurate result.
In the range of 80%abv it is then so inaccurate that the use of a refractometer no longer makes sense.
And on the other side of the reversal point it is unfortunately not ideal either.
A refractometer is therefore more suitable for alcohol solutions when a low alcohol strength is expected.
However, if you have a good balance, you can dilute the solution well weighed before measuring, then measure the now lower alcohol strength and conclude from the measured value to the undiluted alcohol strength. The accuracy of the dilution then determines whether this is of any use. In our calculator Refractometer Temperature Correction Alcohol solutions we have included a calculation option of this dilution method.
Another difference between sugar and alcohol solutions is the range of refractive index:
For sugar, it ranges from 1.333 to 1.3812 (from 0 to 30Brix, which is probably the highest realistic). This is a range of 0.0482.
For alcohol, it ranges from 1.333 to 1.3614 (from 0 - 100% alcohol). This is a range of 0.0284.
So the range of alcohol solutions is much smaller. And this means that the accuracy of the measurement of alcohol solutions is much lower.
However, the range of alcohol solutions is included in the range of sugar solutions. This is different from density meters such as spindles. The density range of alcohol solutions is different from that of sugar solutions. This means that you can use a refractometer for sugar solutions also for alcohol solutions. And the other way around. You only need tables or calculators that can convert the measured values. We have this one for that: Sugar ↔ Refractive index ↔ Alcohol
The temperature correction of refractometer measurements
The "ATC" (automatic temperature compensation) in the inexpensive handheld refractometers works via a bimetal strip. Depending on the temperature, it is bent and the scale is shifted as a result: On the left between the two plastic parts the bimetal strip, on the right the view of the scale.
On the left between the two plastic parts the bimetal strip, on the right the view of the scale.
Since the temperature dependence of the refractive index is different for each substance, the bimatall strip suitable for the substance must be built into the hand refractometer. Since this function cannot be switched off, it is therefore not quite the case that a handheld sugar refractometer could also be used for alcohol. This only works at the calibration temperature of 20°C.
The temperature compensation by a bimetal strip is more or less linear. Or linear to the scale length. This means that it can only work well if the refractive index also behaves just as linearly depending on the temperature and depending on the alcohol or sugar concentration. And that would be a nice coincidence, of course, if it were so. With alcohol solutions, however, the scale itself is not linear at all. And at alcohol strengths above the reversal point, the bimetal strip even corrects in the wrong direction. This automatic temperature compensation is therefore very poor, at least for alcohol solutions. A handheld refractometer without temperature compensation is therefore better for alcohol.
Here are our calculators for temperature correction:
Refractometer Temperature Correction - Sugar solutions
Refractometer Temperature Correction - Alcohol solutions
The adjustment of refractometers
Refractometers can only be adjusted at one point. Normally, one takes the value at water at 20°C. For this we have the calculator Refractometer Adjustment. This means that the further you are from this point, the more uncertain the measurement. And unfortunately, even expensive refractometers are not very accurate. Refractometers have advantages:
- Little liquid is needed.
- The measurement is fast.
- The laboratory instruments have a built-in temperature control. Therefore, the indexes can be measured very quickly at different temperatures.
- Some refractometers can even measure continuously. This means that the changing refractive index of a liquid is measured directly.
But in general the accuracy is lower than with good spindles or other measuring instruments for density, like pycnometers.
So which refractometers are suitable for us?
The bottom line is none. The handheld instruments have too many flaws and the laboratory instruments are too expensive for their only mediocre accuracy. Above all, higher-percentage alcohol solutions should not be measured with refractometers. So the dilution to drinking strength and measurements in the area of the end of the hearts, refractometers are unsuitable for this.
The reason why we nevertheless made so many calculation tools for refractometers is because we simply expected more from them. We had bought old, but actually very good refractometers and repaired them. You can get these old, frequently built models from Zeiss Jena relatively cheap, depending on their condition, but they usually have to be completely disassembled, cleaned, and often a gear has to be replaced: If you like tinkering, then this is a nice project.
However, the accuracy is still not as high as with pycnometers, for example.
If you like tinkering, then this is a nice project.
However, the accuracy is still not as high as with pycnometers, for example.


The second picture shows an Abbe refractometer, a much more expensive professional laboratory instrument. This usually has a scale for the refractive index and one for the sugar content in Brix. However, the alcohol content can also be measured with it. You need a table or calculator for this to infer alcohol strength from refractive index. A temperature compensation is not built in here. This would also be nonsensical, since it is supposed to be a universal measuring instrument, and you would need a different compensation for each substance to be measured. Instead of correcting the measured value depending on the temperature, the measuring head is normally brought to the desired temperature by a temperature-controlled water circuit. Then one is also independent of the availability and accuracy of temperature correction tables or calculators for the respective substance.
The refractive indices of sugar and alcohol solutions
The relationship between refractive index and sugar concentration is quite linear:



However, if you have a good balance, you can dilute the solution well weighed before measuring, then measure the now lower alcohol strength and conclude from the measured value to the undiluted alcohol strength. The accuracy of the dilution then determines whether this is of any use. In our calculator Refractometer Temperature Correction Alcohol solutions we have included a calculation option of this dilution method.
Another difference between sugar and alcohol solutions is the range of refractive index:
For sugar, it ranges from 1.333 to 1.3812 (from 0 to 30Brix, which is probably the highest realistic). This is a range of 0.0482.
For alcohol, it ranges from 1.333 to 1.3614 (from 0 - 100% alcohol). This is a range of 0.0284.
So the range of alcohol solutions is much smaller. And this means that the accuracy of the measurement of alcohol solutions is much lower.
However, the range of alcohol solutions is included in the range of sugar solutions. This is different from density meters such as spindles. The density range of alcohol solutions is different from that of sugar solutions. This means that you can use a refractometer for sugar solutions also for alcohol solutions. And the other way around. You only need tables or calculators that can convert the measured values. We have this one for that: Sugar ↔ Refractive index ↔ Alcohol
The temperature correction of refractometer measurements
The "ATC" (automatic temperature compensation) in the inexpensive handheld refractometers works via a bimetal strip. Depending on the temperature, it is bent and the scale is shifted as a result:

Since the temperature dependence of the refractive index is different for each substance, the bimatall strip suitable for the substance must be built into the hand refractometer. Since this function cannot be switched off, it is therefore not quite the case that a handheld sugar refractometer could also be used for alcohol. This only works at the calibration temperature of 20°C.
The temperature compensation by a bimetal strip is more or less linear. Or linear to the scale length. This means that it can only work well if the refractive index also behaves just as linearly depending on the temperature and depending on the alcohol or sugar concentration. And that would be a nice coincidence, of course, if it were so. With alcohol solutions, however, the scale itself is not linear at all. And at alcohol strengths above the reversal point, the bimetal strip even corrects in the wrong direction. This automatic temperature compensation is therefore very poor, at least for alcohol solutions. A handheld refractometer without temperature compensation is therefore better for alcohol.
Here are our calculators for temperature correction:
Refractometer Temperature Correction - Sugar solutions
Refractometer Temperature Correction - Alcohol solutions
The adjustment of refractometers
Refractometers can only be adjusted at one point. Normally, one takes the value at water at 20°C. For this we have the calculator Refractometer Adjustment. This means that the further you are from this point, the more uncertain the measurement. And unfortunately, even expensive refractometers are not very accurate. Refractometers have advantages:
- Little liquid is needed.
- The measurement is fast.
- The laboratory instruments have a built-in temperature control. Therefore, the indexes can be measured very quickly at different temperatures.
- Some refractometers can even measure continuously. This means that the changing refractive index of a liquid is measured directly.
But in general the accuracy is lower than with good spindles or other measuring instruments for density, like pycnometers.
So which refractometers are suitable for us?
The bottom line is none. The handheld instruments have too many flaws and the laboratory instruments are too expensive for their only mediocre accuracy. Above all, higher-percentage alcohol solutions should not be measured with refractometers. So the dilution to drinking strength and measurements in the area of the end of the hearts, refractometers are unsuitable for this.
The reason why we nevertheless made so many calculation tools for refractometers is because we simply expected more from them. We had bought old, but actually very good refractometers and repaired them. You can get these old, frequently built models from Zeiss Jena relatively cheap, depending on their condition, but they usually have to be completely disassembled, cleaned, and often a gear has to be replaced:
